Flare Spectroscopy Activity
This activity simulates the monitoring of a gas refinery flare with a Do-It-Yourself spectrometer indoors by substituting a candle for the flare.
- Time and participants: The activity should take approximately 1 hour and is suitable for a group of 4, or multiple groups of 4. 1 hour longer if you need to assemble the spectrometer.
- Related research: http://publiclab.org/tag/flare
- You'll need:
- a DIY spectrometer (some require a webcam or smartphone)
- a candle or bunsen burner
- a tripod (tabletop probably best) to stably point the device
- zip ties
- salt & water
- a small spray bottle
- a fluorescent light (the ceiling lights may suffice) for calibration
Preparation
Setup: Be sure you have internet access, or you may use just a webcam program and upload your data later. Fill the spray bottle with very salty water and confirm that you can "mist" the water over the flame without putting it out. A bunsen burner can be substituted for the candle.
Which spectrometer? All of the different versions of the Public Lab spectrometer can be used for this activity. The Desktop Kit can be plugged into a laptop and the spectra graphed in real time, which can be nice for using with a projector, and for viewing the wavelengths in a quantitative way -- but works best with an internet connection, using the https://spectralworkbench.org software. The smartphone version (to be released soon) and the foldable mini spectrometer can be used with a webcam, iPhone, or Android phone, the latter two either with the native camera app or at https://spectralworkbench.org.
Calibration: If you have limited time, you may calibrate the spectrometer ahead of time. Also decide if you'd like the students to build their own -- perhaps the foldable paper versions, which they can take home. Calibration can be a nice way to explore the emission spectra of mercury in fluorescent lamps, as well.
The activity
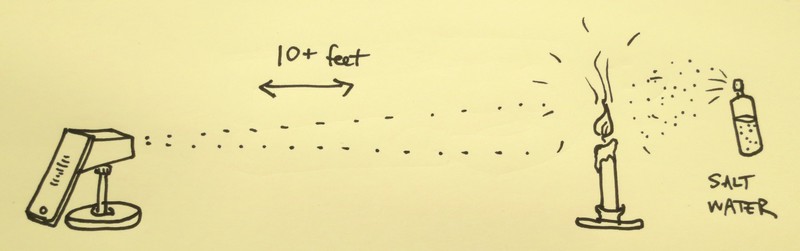
- Set up the candle across the room on a table, light it, and turn off the lights. (see diagram in main image)
- Calibrate the spectrometer. If need be, this can be done ahead of time.
- From the other end of the room, have the students plug in the spectrometer and point it at the flame. This will take some time, as aiming the device may be difficult. (If you are using a Desktop Spectrometry Kit the camera may not be lined up well to point straight out of the device -- it may be biased upwards or downwards)
- Once the flickering candle is lined up and you can see its spectrum, save an image or spectrum (depending on which device you're using).
- Now, mist some salt water over the flame. Observe how the flame turns bright yellow/orange, as in the image above.
Background
Flares from gas refinery smokestacks can reach over 1500 Kelvin, which is hot enough to cause atoms in the flame to produce emission spectra. An explanation of basic spectroscopic theory and links to other documentation is here: http://publiclab.org/wiki/spectrometer-curriculum
By monitoring such flares, Public Lab community researchers hope to identify metals and other contaminants which pose a risk to nearby communities, and to pressure refineries to reduce such pollution.
Public Lab community members using a spectrometer to monitor a real gas refinery flare in Chalmette, Louisiana Read more
Conclusions
At this point, the flare monitoring research is still in progress -- you can follow along and get involved at http://publiclab.org/tag/flare. As a network of collaborators, Public Lab makes progress on technologies and monitoring strategies by working together to improve techniques an innovate new tools step by step, sharing our work on this website.
At this time (Oct 2013), we are continuing to test and improve the flare monitoring equipment and setup, as well as importing data from the NIST database on the spectra for contaminants we expect to identify, for comparison (Read more here) Please share what you do and get in touch with other educators on the plots-education and plots-spectrometry mailing list: http://publiclab.org/wiki/mailing-lists
More resources
- A report from a classroom flame spectroscopy activity using a bunsen burner and various solutions
- Various designs and information about Do-It-Yourself spectrometers
- Recent and ongoing research on flare spectroscopy: http://publiclab.org/tag/flare
- Online spectrometry software and data library: https://spectralworkbench.org
- Overview of spectroscopy with theory and illustrations
](https://i.publiclab.org/system/images/photos/000/001/634/medium/IMG_1794.JPG)
